HIV-1 genotyping and drug resistance mutations in Morocco (2009-2024): a systematic review addressing critical gaps in molecular surveillance
Maryam Ahmina 1, 2, Nada Lamrak 1, 2, Hicham El Annaz 1, 2, 3, Mohamed Rida-Tagagidid 1, 2, 3, Rachid Abi 4, 5, 3, Mohamed Elqatni 1, 2, 3, Abdelilah Laraqi 1, 3, Safae Elkochri 3, Elarbi Bouaiti 2, 3, Ahmed Reggad 1, 3, Youssef Addi 3, Bouchra El Mchichi 1, 3, Nadia Touil 1, 3, Khalid Ennibi 1, 2, 3, Idriss Amine Lahlou 1, 2, 3
1 Molecular Virology and Oncobiology Research Team, Faculty of Medicine and Pharmacy of Rabat, Rabat, Morocco; 2 Faculty of Medicine and Pharmacy, University Mohammed V, Rabat, Morocco; 3 Center of Virology, Infectious and Tropical Diseases, Mohamed V Military Instruction Hospital, Rabat, Morocco; 4 Molecular Virology and Oncobiology Research Team, Faculty of Medicine and Pharmacy of Rabat, Rabat, Morocco; 5 Faculty of Medicine and Pharmacy, University Mohammed V, Rabat, Morocco
*Correspondence: Hicham El Annaz. Email: hichamelannaz74@gmail.com
Received: 24-08-2025
Accepted: 25-10-2025
DOI: 10.24875/AIDSRev.25000022
Available online: 03-12-2025
AIDS Rev. 2026;28(1):1-12
Abstract
The growing use of antiretroviral therapy (ART) has transformed HIV infection into a chronic, manageable disease, yet the emergence of drug resistance continues to threaten global progress. Morocco, located at the crossroads of Sub-Saharan Africa and Europe, offers a unique context for understanding the molecular evolution of HIV in the Middle East and North Africa. This systematic review synthesizes all available data on HIV-1 genotyping and resistance mutations in Morocco from 2009 to 2024, providing the first national overview of molecular resistance patterns. Six studies comprising 673 individuals met the inclusion criteria, spanning 2004-2015. Subtype B predominated (73.8%), followed by CRF02_AG (17.6%), reflecting increasing viral diversification linked to cross-regional transmission. Among ART-experienced patients, acquired drug resistance reached 19.5% at the population level and 53.3% among successfully sequenced samples, with NRTI (48.9%) and PI (22.2%) mutations predominating. The most frequent mutations were M184V (44%), K103N (8.9%), and V82A/L (13.3%). In ART-naïve individuals, transmitted resistance remained limited (1.55%), with no major integrase strand-transfer inhibitor mutations detected, though accessory polymorphisms such as L74M/I and E157Q were present in 3-5% of cases. CD4 counts and viral load suppression improved in later cohorts. These findings underline the critical need to re-establish molecular surveillance in Morocco to capture post-2019 resistance dynamics under dolutegravir-based therapy. Strengthening genotypic monitoring and integrating resistance testing into clinical care will be pivotal to preserving long-term ART efficacy and achieving the UNAIDS 95-95-95 and HIV elimination targets by 2030.
Contents
Introduction
Four decades after its discovery, HIV-1 continues to challenge global health systems, affecting over 40.8 million people living with the virus and resulting in approximately 1.3 million new infections annually as of 20241. The wide-scale practice of antiretroviral therapy (ART) has transformed AIDS into a manageable chronic condition. Currently, over 31.6 million people are receiving ART, which has reduced overall morbidity and mortality. However, the ongoing evolution of HIV-1 presents a significant challenge. The virus’s rapid capacity to adapt and develop resistance to antiretroviral drugs is considered a critical threat to individual treatment outcomes and global control efforts, especially in resource-limited settings2.
In Morocco, HIV was first identified in 1986. Although national prevalence remains low, around 0.1% in the general population, the epidemic is concentrated in key populations. HIV prevalence among men who have sex with men (MSM) rose slightly from 5.1% in 2011 to 5.3% in 2023, whereas it declined among people who inject drugs from 11.4% to 5.3% over the same period. Among migrants, prevalence increased from 3.3% in 2013 to 4.6% in 2022, highlighting persistent vulnerability within mobile groups3–5. Morocco was also the first country in the MENA region to introduce pre-exposure prophylaxis (PrEP), launching a community-based program in 2017 targeting MSM and female sex workers in high-prevalence cities such as Agadir, Casablanca, and Marrakech6. The pilot used daily oral TDF/FTC and achieved 86% uptake and no HIV seroconversions during follow-up, demonstrating the feasibility of PrEP integration within key-population prevention services. Ongoing national expansion seeks to address barriers related to stigma and retention7,8.
As of 2024, an estimated 23,500 people were living with HIV in Morocco, with heterosexual contact remaining the dominant mode of transmission. Since adopting the WHO “Treat All” policy in 2017, Morocco has made substantial progress: 80% of people living with HIV know their status, 95% of those diagnosed receive ART, and 95% of treated individuals have achieved viral suppression6. These achievements illustrate an increasingly effective national response but underscore the importance of closing the remaining gaps in early diagnosis and linkage to care9. In Morocco, ART is delivered through Ministry of Health hospitals and designated reference laboratories, with medications provided free of charge. First-line ART historically used NNRTI-based regimens (ZDV/3TC/EFV or ZDV/3TC/NVP), followed by PI-based second-line combinations (LPV/r or ATV/r). In 2019, the national program incorporated dolutegravir-based first-line therapy (TDF/3TC/DTG) as the preferred first-line regimen, not only for newly diagnosed individuals but also for those already on ART with viral loads (VLs) below 1000 copies/mL, aligning with WHO recommendations and marking a major transition toward integrase (IN) inhibitor-driven ART optimization.
Despite these advances, HIV drug resistance (HIVDR) continues to undermine the durability of ART, particularly in Morocco, where comprehensive molecular data remain scarce10–15. The rapid expansion of ART coverage underscores the need for sustained genotypic surveillance to preserve the efficacy of first- and second-line regimens. Expanding national resistance monitoring is therefore essential to guide treatment adaptation and ensure long-term therapeutic success16.
This review, to our knowledge, synthesizes two decades of evidence on HIV-1 drug resistance in Morocco, focusing on trends in molecular epidemiology, patterns of both acquired drug resistance (ADR) and transmitted drug resistance (TDR), and subtype characteristics. By addressing critical gaps in the current landscape of HIVDR in Morocco and providing actionable insights to update the treatment guidelines, enhance genotyping efforts, and align national data with global initiatives to combat HIV by 2030.
Methods
Search strategy and selection criteria
This review was reported following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines17 (Supplementary Table 1). In this systematic review, we independently searched PubMed, Scopus, and Web of Science to identify relevant studies on HIV-1 drug resistance mutations identified through genotyping in Morocco, published up to November 10, 2024. The search uses a combination of medical subject headings (MeSH) terms and free-text keywords, such as: “HIV-1,” “genotyping,” “drug resistance mutations,” “Morocco,” and “antiretroviral therapy.” Studies were included if they met the following criteria: (i) reported HIV-1 genotyping data from Morocco, (ii) included drug resistance mutation analysis, (iii) involved adult patients (≥ 18 years), (iv) included treatment-naïve and/or experienced patients, and (v) were published as full-text articles in English between 2009 and 2024. Articles were excluded if: (i) they were case reports or case series, (ii) review articles or meta-analyses, (iii) studies without genotypic data, (iv) studies focusing solely on epidemiology or host genetics, (v) conference abstracts, or (vi) studies without clear treatment status information.
Table 1. Descriptive characteristics of the included studies
| Authors and publication | Study design | Study period | Main region | Total sample size | Men, n/N (%) | Median age (years) | Heterosexual transmission (%) | Treatment-status | Median CD4 (cells/mm3) | Median VL (copies/mL) | Mean duration of follow-up (months) | Gene studied | Total samples successfully sequenced | Algorithm |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bakhouch et al., (2009)15 | Cross-sectional | 2004/2007 | Casablanca | 98 | 61/120 (50.8) | 35 | 82.90 | Treatment-naïve | 180 | 114815 | NA | PR/RT | 71 | ANRS/Stanford DB |
| El Annaz et al., (2011)10 | Cross-sectional | 2005/2009 | Multiple regions | 131 | 102/131 (78) | 34 | 89 | Treatment-naïve | 116 | 149050 | NA | PR/RT | 91 | SDRMs |
| El Annaz et al., (2012)11 | Retrospe-ctive cohort | 2005/2010 | Multiple regions | 124 | 116/124 (93.3) | 43 | 88.88 | Treatment-failing | 346 | 70135 | 3-72 | PR/RT | 45 | ANRS |
| Miri et al., (2012)14 | Cross-sectional | 2006/2010 | Multiple regions | 162 | 73/162 (45) | 36 | 82.90 | Treatment-naïve | 270.4 | 169685 | NA | PR | 162 | IAS/ANRS/StanfordDB |
| Alaoui et al., (2018)12 | Cross-sectional | 2009/2015 | Multiple regions | 80 | 62/80 (77.5) | 36 | 81.25 | Treatment-naïve | 409 | 95800 | NA | IN | 77 | StanfordDB |
| Alaoui et al., (2019)13 | Cross-sectional | 2014/2015 | Rabat | 78 | 64/78 (82) | 43 | 92.30 | INSTI-naïve* | 404.5 | 36000 | 7-137 | IN | 78 | StanfordDB |
|
* INSTI-naïve but ART-experienced. NA: not available, PR/RT: protease/reverse transcriptase. |
||||||||||||||
Each article was screened for eligibility first by title and abstract, followed by a full-text assessment. Both screening phases were conducted independently by two researchers, A.M. and L.N., with a third reviewer, E.H., resolving any disagreements. The titles and abstracts were screened using Covidence software, Veritas Health Innovation, Melbourne, Australia. Available at www.covidence.org. for systematic reviews. Data extraction was performed independently by two reviewers using standardized forms in Excel, capturing study characteristics, patient demographics, genotyping methods, and resistance patterns. Any inconsistencies were resolved by consensus with a senior author E.H.
The study is registered with PROSPERO (ID CRD42024619659).
Data analysis
Two independent reviewers extracted data from the included studies using a predefined data extraction form. Discrepancies between reviewers A.M. and L.N. were resolved by a third reviewer E.H.
From the included studies, we extracted the following data using standardized forms in Excel: study characteristics (first author’s name, publication year, study design, study period, setting within Morocco, sample size), patient demographics (gender, age, route of transmission), treatment status (naïve or experienced), clinical parameters (median CD4 count, median VL, mean duration of follow up when reported), genotyping methods used. For resistance data, we recorded the total of successfully sequenced samples, presence and prevalence of major and minor mutations by drug class (nucleoside reverse transcriptase inhibitors [NRTIs], non-nucleoside reverse transcriptase inhibitors [NNRTIs], PIs, integrase strand-transfer inhibitors [INSTIs]), and viral subtypes data. Overall ADR and TDR were computed using the full numbers of ART-experienced and ART-naïve patients, respectively; in contrast, mutation- and class-specific prevalences were calculated only among samples with successful sequencing of the relevant gene.
Study quality was assessed using the Newcastle-Ottawa scale. This scale evaluates studies across three key domains: Selection, comparability, and outcome. Based on established criteria, we classified studies as having low risk of bias (8 or 9 stars), moderate risk (5-7 stars), or high risk (> 5 stars).
Primary outcomes included (i) prevalence of drug resistance mutations by class, (ii) distribution of resistance by treatment status, (iii) HIV-1 subtype distribution and temporal trends, and (iv) virologic and immunologic characteristics. For continuous outcomes, we extracted frequencies and proportions. For categorical outcomes, we documented the presence or absence of specific mutations and resistance patterns. Given the limited number of studies and heterogeneity in reporting, a meta-analysis was not conducted. Instead, we performed a narrative synthesis, organizing data chronologically and by treatment context. Drug resistance data were analyzed descriptively and expressed as proportions. For overall ADR and TDR, prevalence was calculated at two complementary levels. Study-level prevalence corresponded to the proportion of patients with at least one drug resistance mutation among those successfully genotyped within a given cohort, while population-level prevalence represented the proportion of resistant individuals among all participants in each ART status group (ART-experienced or ART-naïve), regardless of sequencing success. For class-specific analyses (NRTI, NNRTI, PI, and INSTI), prevalence was determined within each ART group using only samples successfully sequenced for the relevant gene region (protease [PR], reverse transcriptase [RT], or IN). All results are presented as percentages reflecting the proportion of resistant cases among interpretable sequences within each treatment category. Calculations were performed manually using Microsoft Excel, with results presented in graphs and narrative descriptions. Statistical significance was set at p < 0.05 where applicable.
Results
Study characteristics and design
We identified 107 articles, of which 43 were removed as duplicates. After screening titles and abstracts, 58 full texts were retrieved, and we included six studies in our analysis that met the inclusion criteria (Fig. 1). The main reason for exclusion was the absence of information about the outcomes of interest of our systematic review. The list of excluded references is in supplementary table 2.
Figure 1. Preferred reporting items for systematic reviews and meta-analyses flowchart of the systematic review search strategy.
The six included studies were conducted between 2004 and 2015 and published between 2009 and 2019. Notably, no study on HIV-1 genotypic resistance in Morocco has been published since 2019, highlighting a critical lack of recent molecular surveillance between 2016 and 2024. The six studies covered 673 patients drawn from both single- and multi-regional cohorts. Sample sizes ranged from 78 to 162 participants. All study populations were composed almost exclusively of Moroccan nationals, with no indication of non-Moroccan inclusion. Among these, four studies included ART-naïve patients, whereas two studies focused on ART-experienced individuals; of them, one included INSTI-naïve patients. Genotyping analyses targeted the PR/RT or IN genes, employing validated algorithms such as the ANRS algorithm, StanfordDB, and IAS. All studies used the Capillary sequencing method (Table 1).
Demographic and epidemiological features
The median age of participants ranged from 34 to 43 years, with a male predominance observed in all cohorts (45%-93%). Heterosexual transmission was identified as the primary route of infection in 81-92% of cases. Median baseline CD4 counts varied significantly, with ART-naïve patients showing a median of 116-409 cells/mm3, whereas ART-experienced individuals had a median of 346-404.5 cells/mm3. VL values were highly variable, with ART-naïve cohorts exhibiting median VLs up to 169,685 copies/mL, whereas ART-experienced patients had a median VL of 36,000-114,815 copies/mL. More recent studies (2014-2015) reported higher median CD4 counts (404.5 cells/mm3) and lower VLs, suggesting improved immune status over time (Table 1).
HIV-1 subtype distribution and evolution (2004-2015) in Morocco
Across all 524 genotyped samples collected between 2004 and 2015, subtype B remained the dominant form (73.8%), followed by CRF02_AG (17.6%), confirming a dual epidemic pattern shaped by both Western and sub-Saharan introductions. Minor subtypes were less frequent, including A1 (5.4%), C (1.6%), and other recombinant forms (3.6%), which together represented fewer than 10% of all sequences (Fig. 2A).
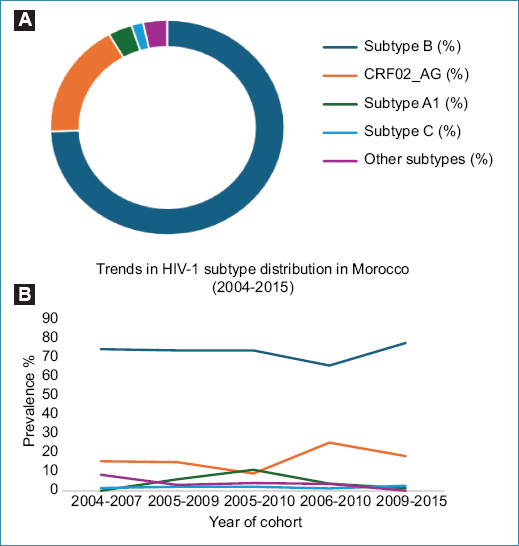
Figure 2. Distribution and trends of HIV-1 subtypes in Morocco (2004-2015). A: the donut chart highlights the distribution of HIV-1 subtypes in Morocco between 2004 and 2015. B: illustrates temporal trends in subtype prevalence.
Temporal comparison across study periods revealed that subtype B remained the dominant strain, with stabilization and then a slight increase from 74.6% in 2004-2007 to 77.92% in 2009-2015. In contrast, CRF02_AG prevalence fluctuated but ultimately increased, rising from 15.5% to 18.18% over the same period, reflecting a gradual diversification of the epidemic. Meanwhile, subtypes A1, C, and other recombinant forms showed a decreasing trend, with A1 dropping from 6% to 1.3%, C fluctuating between 1.4% and 2.6%, and other minor subtypes declining from 8.4% to 0% (Fig. 2B).
Drug resistance patterns
The overall prevalence of drug resistance was markedly higher among ART-experienced individuals (19.5% at the population level 24/123) compared with ART-naïve patients (1.5%, 7/479), confirming the cumulative selective pressure imposed by prolonged antiretroviral exposure. The distribution of ADR and TDR across ART-naïve and ART-experienced cohorts is illustrated in figure 3. When restricted to the study that performed genotyping among experienced individuals11, the study-level prevalence of ADR reached 53.3% (24 of 45 sequenced), underscoring the substantial burden of resistance among successfully genotyped patients. In contrast, TDR among ART-naïve individuals remained very low, estimated at 4.3% (7 of 162 genotyped) within PR/RT-sequenced cohorts.
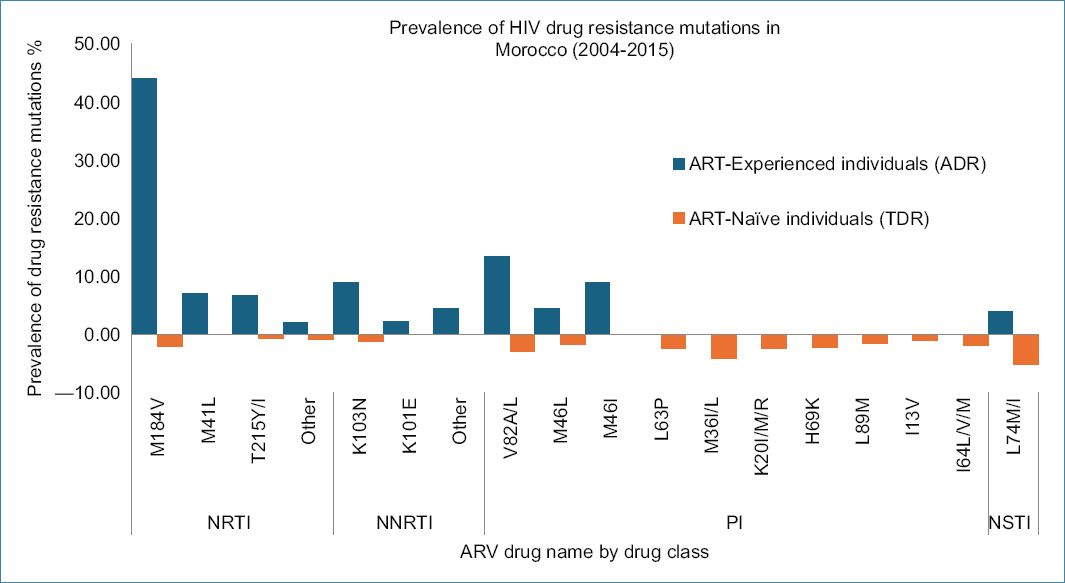
Figure 3. Estimated prevalence of HIV-1 drug resistance mutations in ART-naive and ART-experienced individuals in Morocco between 2004 and 2015. ARV: antiretroviral drugs; NRTI: nucleoside reverse transcriptase inhibitors; NNRTI: non-nucleoside reverse transcriptase inhibitors; PI: protease inhibitors; INSTI: integrase strand transfer inhibitors.
Among ART-experienced participants, NRTI resistance predominated (48.9%, 22 of 45 sequenced), followed by PI resistance (22.2%, 10 of 45) and NNRTI resistance (13.3%, 6 of 45). In ART-naïve individuals, class-specific resistance remained limited with NRTI (2.5%, 4 of 162 genotyped), NNRTI (2.5%, 4 of 162), and PI (1.23%, 4 of 324). IN inhibitor (INSTI) resistance was absent in both groups, with only accessory polymorphisms detected among INSTI-naïve patients (5.2% of 155).
NRTI and NNRTI resistance
NRTI resistance mutations were the most frequent among ART-experienced individuals, detected in 48.9% (22/45) of genotyped cases. The M184V substitution was predominant (44.4%, 20 of 45), conferring high-level resistance to lamivudine and emtricitabine. Thymidine-analog mutations (TAMs) such as M41L (6.7%) and T215Y/I (6.7%) were also common, reflecting prolonged exposure to zidovudine-based regimens. In ART-naïve individuals, M184V was infrequent (1.85%, 3/162) and T215I/S occurred in 1.23% (2/162), confirming minimal transmission of NRTI-associated resistance mutations.
NNRTI resistance was detected in 13.3% (6/45) of ART-experienced patients, mainly due to K103N (8.9%, 4/45), which confers high-level resistance to efavirenz and nevirapine, and K101E (2.2%, 1/45), associated with reduced susceptibility to first-generation NNRTIs. In ART-naïve individuals, K103N remained the only detected NNRTI mutation (2.5%, 4/162), emphasizing that transmitted NNRTI resistance was rare during the 2004-2015 period. These findings reflect the historical reliance on NNRTI-based first-line regimens.
Protease inhibitor (PI) resistance
PI resistance was identified in 22.2% (10/45) of ART-experienced individuals, underscoring the selective impact of PR-based regimens on viral evolution. Major PI-associated mutations in experienced patients included V82A/L (13.3%, 6/45), M46I (8.8%, 4/45), and M46L (4.4%, 2/45), all of which reduce susceptibility to ritonavir-boosted atazanavir, lopinavir, and indinavir. In ART-naïve individuals, major PI mutations were uncommon, limited to M46L (0.61%, 2/324) and V82L/I (0.61%, 2/324) and a single V82A (0.31%, 2/324), yielding a pooled prevalence of 1.54% (5/324) across naïve cohorts. These mutations likely represent isolated transmission events rather than widespread dissemination of PI-resistant strains.
PI-associated substitutions in naïve patients were otherwise predominantly polymorphic variants, including M36I/L, L63P, H69K, L89M, I13V, K20I/M/R, and I64L/V/M, occurring at frequencies between 4% and 6%. These polymorphisms do not confer significant loss of PI susceptibility but form a genetic background that may facilitate the emergence of resistance under therapeutic pressure, reinforcing the need for continued molecular surveillance.
INSTI resistance
IN strand transfer inhibitor resistance mutations were infrequent across all cohorts with no major mutations detected in either ART-experienced or ART-naïve individuals, accessory mutations, such as L74M/I, E157Q, and T97A, were found in 3.84% of ART-experienced and 5.2% of ART-naïve cases, highlighting the potential for resistance evolution under therapeutic pressure, particularly as INSTIs become more widely used in Morocco.
Subtype-specific resistance patterns
Across Moroccan studies assessing subtype-resistance associations11,14,15, subtype B remained predominant, but non-B variants, including CRF02_AG, A1, G, and C, showed distinct PR polymorphism profiles. In El Annaz (2012)11, overall resistance was comparable between subtype B (54%) and non-B (50%), yet dual NRTI-PI resistance occurred more often among non-B viruses. Among these, M184V was detected across both subtype groups, whereas M46I/L and V82A/L were mostly confined to subtype B sequences. Non-B viruses, particularly CRF02_AG and A1, carried a dense background of PR polymorphisms such as M36I, H69K, L89M, and K20I/M/R present in nearly all non-B isolates but rare in subtype B.
Among treatment-naïve individuals (Bakhouch 2009; Miri 2012), major PI mutations such as M46L and V82L/A were again observed exclusively in subtype B (≈ 4%), whereas non-B subtypes displayed a markedly higher prevalence of natural PI-region polymorphisms (M36I/L, K20I, H69K, I13V, L63P) affecting over 70% of sequences.
Although major drug-resistance mutations occurred at similar frequencies between subtype B and non-B viruses, the extensive polymorphic landscape of non-B strains may alter the genetic pathways leading to PI resistance.
Antiretroviral treatment regimens and duration across cohorts
ART regimens varied across cohorts, with combinations such as zidovudine + lamivudine + efavirenz (ZDV + 3TC + EFV) and zidovudine + lamivudine + indinavir (ZDV + 3TC + IND) commonly reported. Treatment durations ranged from 3 months to over 11 years for long-term ART-experienced patients.
Study quality and risk of bias assessment
Overall, all studies achieved high to medium-quality scores (7-9 stars), indicating a low risk of bias (≥ 8 stars). A detailed risk of bias assessment can be found in supplementary table 3.
Discussion and implications
This review provides the first comprehensive synthesis of HIV-1 drug resistance in Morocco, revealing a dual challenge of emerging subtype diversification and persistent NRTI/NNRTI resistance under historical treatment regimens. It consolidates evidence from six studies conducted between 2004 and 2015, with publication dates extending to 2019. Notably, no Moroccan HIV-1 genotyping publications were identified after 2015 despite a comprehensive search through 2024, underscoring a critical 9-year gap in molecular surveillance. This hiatus highlights the urgent need to reintegrate genotypic monitoring into national HIV programs. The six studies collectively analyzed 673 patients and provided valuable insights into the molecular epidemiology of HIV-1 in Morocco and its therapeutic implications. The findings emphasize significant drug resistance patterns among both ART-naïve and ART-experienced individuals, shaped by the interaction of viral subtypes, treatment practices, and regional epidemiological context.
Subtype distribution and global comparisons
The rise of CRF02_AG alongside subtype B’s persistence suggests a growing influence of non-B recombinant forms, aligning with the shift toward sub-Saharan HIV-1 genetic patterns hypothesis of HIV-1 molecular evolution in Morocco. This shift may be driven by increased migration and cross-border transmission from sub-Saharan Africa, where CRF02_AG is highly prevalent, likely driven by the country’s strategic position at the nexus of Europe and Africa. The continued dominance of subtype B alongside the emergence of CRF02_AG highlights the evolving genetic landscape of HIV-1 in Morocco, warranting further surveillance and molecular epidemiology studies to assess the potential impacts of ART efficacy and resistance patterns18.
These trends, observed in Moroccan cohorts from 2004 to 2015, marked a divergence from the broader MENA region, where subtype B remained predominant in countries such as Algeria19 and Tunisia20. However, during this period, Morocco’s higher prevalence of CRF02_AG aligned it more closely with patterns in West and Central Africa, where this strain was the most common21. This recombinant form was more prevalent in other MENA countries, such as Egypt22, and less prevalent in the Gulf Cooperation Council countries, where subtype C dominated, particularly among expatriate populations23.
Morocco’s subtype profile between 2004 and 2015 also differed from the patterns observed in other regions of Africa, where subtype C overwhelmingly dominated Southern Africa24,25, and subtype A was the most prevalent in Eastern Africa26. These subtypes were associated with rapid disease progression and specific resistance mutations, creating distinct challenges for ART effectiveness. By contrast, the greater subtype diversity in West and Central Africa, including CRF02_AG27,28, CRF01_AE, and CRF06_cpx21, was reflected in Morocco’s epidemiological landscape during this period, further emphasizing the country’s connectivity with these regions18.
The data from 2004 to 2015 showed that Morocco’s subtype B predominance more closely mirrored European epidemiological trends, where subtype B accounts for most cases due to its early introduction and spread during the initial waves of the epidemic29–33. This consistency has shaped global research and treatment protocols, as subtype B has been the focus of most clinical trials and drug development efforts. However, in the last few years, the increasing prevalence of non-B subtypes in Europe, including CRF02_AG and F1 in Spain34,35, sub-subtype F1 in Belgium36, CRF60_BC in Italy37, CRF06_cpx in Southwest France38, alongside subtype B, reflects the impact of migration from Africa and Asia contributing to greater genetic diversity within the European HIV epidemic39,40. Given Morocco’s position at the crossroads of Europe, North Africa, and sub-Saharan Africa, it is highly likely that these evolving subtype distributions are influencing the Moroccan HIV epidemic as well. Comprehensive genotyping efforts are urgently needed to determine whether Morocco is experiencing similar shifts in subtype diversity as neighboring regions and to ensure that national ART guidelines remain aligned with the true epidemiological landscape of the country.
Resistance patterns across regions
The resistance patterns in Moroccan cohorts from 2004 to 2015 reflect both regional and global trends. These resistance profiles must be interpreted in the context of Morocco’s evolving ART landscape. Early cohorts were treated with NNRTI-based regimens such as ZDV/3TC/EFV or ZDV/3TC/NVP, whereas since 2019, dolutegravir-based combinations (TDF/3TC/DTG) have become the national first-line standard. This regimen shift, initiated shortly after the WHO’s “Treat All” recommendation in 2017, marked a turning point toward higher-barrier INSTI-based therapy and likely contributed to the decline of NNRTI-associated resistance41.
Among ART-naïve individuals, the weighted prevalence of TDR was estimated at 1.55% at the population level (7/479) and 4.3% among successfully sequenced PR/RT samples (7/162), with most resistance mutations affecting NRTIs and NNRTIs. The M184V mutation was detected in 1.85% of ART-naïve individuals, indicating a low but notable level of resistance to lamivudine, whereas TAMs, such as T215Y/I, remained rare (1.23%). NNRTI resistance mutations were also limited, with K103N detected in 2.5% of cases, highlighting a controlled spread of resistance despite historical reliance on NNRTI-based regimens42.
Compared to other regions, Morocco’s TDR rates were relatively low. This contrasts sharply with sub-Saharan Africa, where TDR rates often exceed 10%, particularly in high-burden countries, where early reliance on NNRTI-based regimens has contributed to widespread resistance43–47. Similarly, in Europe, TDR rates in countries with long-established ART programs, such as France (10.8%), Spain (8.3%), and Italy (7.3%), are significantly higher, reflecting the longer duration and broader rollout of ART, which has facilitated the spread of resistant strains48–52. The predominance of mutations such as K103N in these regions highlights the challenges of managing resistance in NNRTI-heavy treatment protocols53. However, the limited availability of studies on TDR prevalence in the MENA region, including Tunisia, Algeria, Egypt, and the Gulf region, makes direct comparisons challenging, likely due to later ART rollouts and restricted drug distribution policies54. This lack of data highlights a major gap in HIV molecular surveillance in the region, reinforcing the need for updated resistance studies in Morocco to assess whether recent treatment expansions and subtype shifts have influenced resistance patterns.
Among ART-experienced individuals in Morocco, the overall prevalence of ADR was significantly higher, reaching 19.5% at the population level (24/123) and 53.3% among successfully sequenced PR/RT samples (24/45). Class-specific resistance showed NRTIs affected in 48.9% (22/45), NNRTIs in 13.3% (6/45), and PIs in 22.2% (10/45). This finding aligns with global patterns observed in resource-limited settings, where challenges such as treatment adherence, delayed VL monitoring, and limited routine resistance testing exacerbate the development of resistance mutations over time55. M184V (44%) remained the most frequent NRTI resistance mutation, followed by M41L (6.7%) and T215Y/I (6.7%), reflecting cumulative exposure to zidovudine-based regimens and lamivudine. For NNRTIs, K103N (8.88%) was the most prevalent mutation, while K101E was detected at lower rates (2.22%) but still contributed to cross-resistance within the NNRTI drug class. PI resistance was primarily V82A/L (13.33%) and M46I/L (8.88%), whereas minor PI polymorphisms, such as L63P, M36I/L, and K20I/M/R, were common in ART-naïve individuals, suggesting a background of natural variation rather than drug-induced resistance. However, when combined with major PI mutations, these minor mutations could enhance viral fitness and resistance development.
Data from neighboring countries in the MENA region remains limited56. Resistance patterns observed in Morocco between 2004 and 2015 closely resemble trends in high-burden settings such as South Africa and Kenya. ADR rates often exceeded 25%, with widespread M184V and NNRTI resistance mutations such as K103N57,58. The higher ADR rates in these regions highlight the impact of delayed regimen optimization and limited adherence support. In contrast, European countries with long-established ART programs report lower ADR rates, primarily due to routine VL monitoring and widespread access to resistance testing59,60. Despite this, M184V remains one of the most commonly reported mutations globally, reflecting the continued reliance on lamivudine in first-line ART regimens61.
Interpretation of IN resistance in Morocco is constrained by the time window of the available evidence (2004-2015), which predates the 2019 rollout of dolutegravir-based ART. Across the two IN studies, all participants were INSTI-naïve, and no major INSTI resistance-associated mutations were detected. Only accessory substitutions, L74M/I, E157Q, and T97A, were observed at low frequencies (3.84% in ART-experienced and 5.2% in ART-naïve cohorts), consistent with natural polymorphisms rather than drug-selected resistance. These data therefore represent baseline IN variability before INSTI exposure. Prospective, post-2019 surveillance, particularly genotyping at virological failure on dolutegravir, is needed to detect any emergence of INSTI resistance under the national program.
Overall, Morocco’s comparatively low prevalence of NNRTI, PI, and INSTI resistance likely reflects both shorter ART exposure and the proactive adoption of high-barrier regimens. Regional evidence suggests that moderate resistance levels across MENA countries are largely due to delayed ART rollout followed by a rapid transition toward modern regimens with stronger resistance barriers. Continued genotypic monitoring will be essential to preserve these favorable resistance trends and sustain the long-term efficacy of national treatment programs54.
Resistance patterns in Morocco have evolved significantly over time, with early cohorts (2004-2007) showing sporadic cases of TDR, particularly NNRTI mutations such as K103N, which declined in later years as higher resistance-barrier regimens replaced older NNRTI-based therapies. Among ART-experienced individuals, resistance rates for NRTIs, NNRTIs, and PIs remained persistently high, driven by prolonged ART exposure and virological failure. Notably, minor PI mutations such as L63P and M36I/L remained stable but showed a slight decline in later cohorts (2014-2015), reflecting improvements in treatment adherence and earlier ART initiation.
Our findings reveal limited differences in the overall prevalence of major drug-resistance mutations between subtype B and non-B viruses circulating in Morocco, yet clear contrasts in PR gene diversity. In line with observations from other regions of the Middle East and North Africa, non-B subtypes particularly CRF02_AG, A1, and G displayed a substantially higher frequency of natural polymorphisms such as M36I, H69K, L89M, and K20I/M/R, which, although not conferring resistance on their own, can modulate the genetic barrier to PI and facilitate compensatory adaptations once selective pressure is applied. Conversely, major PI mutations (M46I/L, V82A/L) were largely confined to subtype B, consistent with the longer historical exposure of subtype B viruses to first-generation PI in treated populations. Similar patterns have been reported in North and West African cohorts, where non-B variants often exhibit higher baseline variability but fewer major protease mutations under limited ART exposure. These findings highlight the importance of integrating subtype-specific molecular surveillance into routine genotyping and resistance monitoring programs, as Morocco’s epidemic continues to diversify through the growing circulation of recombinant forms.
Despite these advances, our understanding of HIV-1 in Morocco remains incomplete. Studies conducted between 2004 and 2015 suggest that HIV-1 subtype B was the most common, but over time, the presence of CRF02_AG, a recombinant form typically found in West and Central Africa, began to increase. Minor subtypes such as A1, C, and other recombinant strains were also detected, though at lower frequencies. This shift in Morocco’s HIV genetic diversity suggests a growing influence of migration and regional connectivity with sub-Saharan Africa, making it even more critical to understand how these changes might affect treatment outcomes and resistance patterns today. Unfortunately, there have been no large-scale molecular studies in recent years, between 2016 and 2024, to track how these subtypes may have continued to evolve. This lack of updated data extends to HIVDR as well. While earlier studies provided some insights into TDR and ADR, most were limited in scope, focusing on small sample sizes and partial gene sequencing. Routine HIV genotyping is not yet integrated into clinical care, and Morocco has yet to conduct a national drug resistance survey in line with WHO recommendations. Meanwhile, resistance to older drug classes, especially NNRTIs, has continued to rise globally, and IN inhibitors, now the cornerstone of HIV treatment, are being used more widely than ever. Without updated surveillance, it remains unclear whether Moroccan patients receiving ART today are benefiting from the most effective regimens available.
These gaps in knowledge between 2016 and 2024 highlight the urgent need for renewed molecular surveillance efforts. As drug resistance, treatment failure, and viral evolution continue to shape the epidemic, ensuring that Moroccan HIV research keeps pace with global developments is crucial; not just for improving individual patient outcomes, but for guiding public health policies, treatment strategies, and future HIV prevention efforts.
To sustain the efficacy of HIV treatment strategies and adapt to the evolving epidemic, Morocco must prioritize comprehensive molecular surveillance, integrate routine resistance testing into clinical practice, and refine ART protocols based on real-time epidemiological data. Strengthening national genotyping efforts and optimizing first-line and salvage regimens will be essential in mitigating the spread of drug-resistant strains and preserving long-term treatment success. This will further enhance therapeutic durability and contribute to the broader goal of achieving sustained AIDS epidemic elimination by 2030.
Conclusion
HIV-1 drug resistance in Morocco remains relatively limited but shows clear signals of evolutionary change driven by shifting treatment practices and expanding subtype diversity. The absence of recent genotyping studies, particularly in the dolutegravir era, represents a critical gap in national surveillance. Re-establishing routine resistance testing, integrating subtype-specific monitoring, and strengthening laboratory capacity aligned with WHO 2030 objectives are essential to preserve the durability of current regimens. Robust molecular surveillance will be central to safeguarding long-term treatment effectiveness and guiding future HIV policy in Morocco
Author contributions
M. Ahmina, H. El Annaz, K. Ennibi, and I. Amine Lahlou: conceptualization and study design. M. Ahmina: study design. M. Ahmina and N. Lamrak: systematic literature search. M. Ahmina, M. Rida-Tagagidid, R. Abi, A. Laraqi, and E. Bouaiti: methodology. M. Ahmina, N. Lamrak, and H. El Annaz: data extraction. A. Laraqi: software. M. Ahmina and N. Lamrak: quality assessment. H. El Annaz, A. Reggad, N. Touil, K. Ennibi: validation. N. Lamrak, R. Abi, M. Elqatni, B. El Mchichi, N. Touil: formal analysis. M. Ahmina. H. El Annaz, A. Reggad: investigation. A. Reggad, M. Elqatni, Y. Addi: Resources; M. Ahmina and Y. Addi: data curation. M. Ahmina: writing–original draft preparation. S. Elkochri: review and editing. M. Ahmina: visualization. H. El Annaz: supervision. K. Ennibi and I. Amine Lahlou: project administration. All authors have read and agreed to the published version of the manuscript.
Funding
None.
Conflicts of interest
None.
Ethical considerations
Protection of human subjects and animals. The authors declare that no experiments on humans or animals were performed for this research.
Confidentiality, informed consent, and ethical approval. This study does not involve personal patient data, medical records, or biological samples, and does not require ethical approval. SAGER guidelines do not apply.
Declaration on the use of artificial intelligence (AI). The authors declare that no generative artificial intelligence was used in the writing or creation of the content of this manuscript.
Supplementary data
Supplementary data are available at DOI: 10.24875/AIDSRev.25000022. These data are provided by the corresponding author and published online for the benefit of the reader. The contents of supplementary data are the sole responsibility of the authors.
References
1. World Health Organization. HIV Statistics, Globally and by WHO Region, 2024. Geneva:World Health Organization;2024.
2. Pennings PS. HIV drug resistance:problems and perspectives. Infect Dis Rep. 2013;5:5.
3. Global HIV &AIDS Statistics – Fact Sheet. UNAIDS. Available from:https://www.unaids.org/en/resources/fact-sheet [Last accessed on 2025 Apr 13].
4. ALCS Maroc. Rapport D'activite ALCS 2023. Maroc:Available from:https://alcs.ma/wp-content/uploads/2024/07/rapport-dactivite-alcs-2023-version-def.pdf
5. Rapport National SIDA 2024 Maroc. Morocco:Ministre de la Santéet de la Protection Sociale;2024. Available from:https://www.sante.gov.ma/Documents/2024/11/Rapport%20SIDA%202024%20VF%20.pdf [Last accessed on 2025 Apr 01].
6. Ben Moussa A, Belhiba O, Sodqi M, Hajouji FZ, Salah NE, Sakhri N, et al. PrEPare_Morocco a successful community-based PrEP delivery demonstration program for men who have sex with men and female sex workers in Morocco. AIDS Care. 2024;36:508-16.
7. Les Outils de Prévention – ALCS. Available from:https://alcs.ma/les-outils-de-prevention-2 [Last accessed on 2025 Oct 22].
8. Moussa AB, Badahdah AM, Hidous K, Barakad R, Diallo F, TraoréM, et al. Barriers to Oral PrEP:a qualitative study of female sex workers, PrEP prescribers, policymakers, and community advocates in Morocco. J Int Assoc Provid AIDS Care. 2024;23:23259582241266691.
9. Maroc ONUSIDA. Available from:https://www.unaids.org/fr/regionscountries/countries/morocco [Last accessed on 2025 Jan 01].
10. Annaz HE, Recordon-Pinson P, Baba N, Sedrati O, Mrani S, Fleury H. Presence of drug resistance mutations among drug-naive patients in Morocco. AIDS Res Hum Retroviruses. 2011;27:917-20.
11. El Annaz H, Recordon-Pinson P, Tagajdid R, Doblali T, Belefquih B, Oumakhir S, et al. Drug resistance mutations in HIV type 1 isolates from patients failing antiretroviral therapy in Morocco. AIDS Res Hum Retroviruses. 2012;28:944-8.
12. Alaoui N, El Alaoui MA, Touil N, El Annaz H, Melloul M, Tagajdid R, et al. Prevalence of resistance to integrase strand-transfer inhibitors (INSTIs) among untreated HIV-1 infected patients in Morocco. BMC Res Notes. 2018;11:369.
13. Alaoui N, El Alaoui MA, El Annaz H, Farissi FZ, Alaoui AS, El Fahime E, et al. HIV-1 integrase resistance among highly antiretroviral experienced patients from Morocco. Intervirology. 2019;62:65-71.
14. Miri L, Ouladlahsen A, Kettani A, Bensghir R, Marhoum Elfilali K, Wakrim L. Characterization of protease resistance-associated mutations in HIV type 1 drug-naive patients following the increasing prevalence of the CRF02-AG strain in Morocco. AIDS Res Hum Retroviruses. 2012;28:571-7.
15. Bakhouch K, Oulad-Lahcen A, Bensghir R, Blaghen M, Elfilali KM, Ezzikouri S, et al. The prevalence of resistance-associated mutations to protease and reverse transcriptase inhibitors in treatment-naïve (HIV1)-infected individuals in Casablanca, Morocco. J Infect Dev Ctries. 2009;3:380-91.
16. Consolidated Guidelines on the Use of Antiretroviral Drugs for Treating and Preventing HIV Infection:Recommendations for a Public Health Approach. 2nd ed. Available from:https://www.who.int/publications/i/item/9789241549684 [Last accessed on 2025 Jan 01].
17. Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses:the PRISMA statement. Int J Surg. 2010;8:336-41.
18. Miri L, Wakrim L, Kassar H, Hemminki K, Khyatti M. Impact of immigration on HIV-1 molecular epidemiology in West Africa, Maghreb and southern Europe. AIDS Rev. 2014;16:109-16.
19. Bouzeghoub S, Jauvin V, Recordon-Pinson P, Garrigue I, Amrane A, Belabbes EH, et al. High diversity of HIV type 1 in Algeria. AIDS Res Hum Retroviruses. 2006;22:367-72.
20. El Moussi A, Thomson MM, Delgado E, Cuevas MT, Nasr M, Abid S, et al. Genetic diversity of HIV-1 in Tunisia. AIDS Res Hum Retroviruses. 2017;33:77-81.
21. Hemelaar J, Elangovan R, Yun J, Dickson-Tetteh L, Fleminger I, Kirtley S, et al. Global and regional molecular epidemiology of HIV-1, 1990–2015:a systematic review, global survey, and trend analysis. Lancet Infect Dis. 2019;19:143-55.
22. Amer AN, Gaballah A, Emad R, Ghazal A, Attia N. Molecular epidemiology of HIV-1 virus in Egypt:a major change in the circulating subtypes. Curr HIV Res. 2021;19:448-56.
23. Badreddine S, Smith K, Van Zyl H, Bodelle P, Yamaguchi J, Swanson P, et al. Identification and characterization of HIV type 1 subtypes present in the kingdom of Saudi Arabia:high level of genetic diversity found. AIDS Res Hum Retroviruses. 2007;23:667-74.
24. Sivay MV, Hudelson SE, Wang J, Agyei Y, Hamilton EL, Selin A, et al. HIV-1 diversity among young women in rural South Africa:HPTN 068. PLoS One. 2018;13:0198999.
25. Ngcapu S, Theys K, Libin P, Marconi VC, Sunpath H, Ndung'u T, et al. Characterization of nucleoside reverse transcriptase inhibitor-associated mutations in the RNase H region of HIV-1 subtype C infected individuals. Viruses. 2017;9:330.
26. Gounder K, Oyaro M, Padayachi N, Zulu TM, de Oliveira T, Wylie J, et al. Complex subtype diversity of HIV-1 among drug users in major Kenyan cities. AIDS Res Hum Retroviruses. 2017;33:500-10.
27. Lihana RW, Ssemwanga D, Abimiku A, Ndembi N. Update on HIV-1 diversity in Africa:a decade in review. AIDS Rev. 2012;14:83-100.
28. Nii-Trebi NI, Brandful JA, Ibe S, Sugiura W, Barnor JS, Bampoh PO, et al. Dynamic HIV-1 genetic recombination and genotypic drug resistance among treatment-experienced adults in northern Ghana. J Med Microbiol. 2017;66:1663-72.
29. Sallam M, Esbjörnsson J, Baldvinsdóttir G, Indriðason H, Björnsdóttir TB, Widell A, et al. Molecular epidemiology of HIV-1 in Iceland:early introductions, transmission dynamics and recent outbreaks among injection drug users. Infect Genet Evol. 2017;49:157-63.
30. Tumiotto C, Bellecave P, Recordon-Pinson P, Groppi A, Nikolski M, Fleury H. Diversity of HIV-1 in aquitaine, Southwestern France, 2012-2016. AIDS Res Hum Retroviruses. 2018;34:471-3.
31. Volz EM, Le Vu S, Ratmann O, Tostevin A, Dunn D, Orkin C, et al. Molecular epidemiology of HIV-1 subtype B reveals heterogeneous transmission risk:implications for intervention and control. J Infect Dis. 2018;217:1522-9.
32. Hebberecht L, Vancoillie L, Schauvliege M, Staelens D, Dauwe K, Mortier V, et al. Frequency of occurrence of HIV-1 dual infection in a Belgian MSM population. PLoS One. 2018;13:0195679.
33. Alexiev I, Lo Presti A, Dimitrova R, Foley B, Gancheva A, Kostadinova A, et al. Origin and spread of HIV-1 subtype B among heterosexual individuals in Bulgaria. AIDS Res Hum Retroviruses. 2018;34:244-53.
34. Delgado E, Benito S, Montero V, Cuevas MT, Fernández-García A, Sánchez-Martínez M, et al. Diverse large HIV-1 non-subtype B clusters are spreading among men who have sex with men in Spain. Front Microbiol. 2019;10:655.
35. Kostaki EG, Flampouris A, Karamitros T, Chueca N, Alvarez M, Casas P, et al. Spatiotemporal characteristics of the largest HIV-1 CRF02_AG outbreak in Spain:evidence for onward transmissions. Front Microbiol. 2019;10:370.
36. Vinken L, Fransen K, Cuypers L, Alexiev I, Balotta C, Debaisieux L, et al. Earlier initiation of antiretroviral treatment coincides with an initial control of the HIV-1 sub-subtype F1 outbreak among men-having-sex-with-men in Flanders, Belgium. Front Microbiol. 2019;10:613.
37. Lai A, Simonetti FR, Brindicci G, Bergna A, Di Giambenedetto S, Sterrantino G, et al. Local epidemics gone viral:evolution and diffusion of the Italian HIV-1 recombinant form CRF60_BC. Front Microbiol. 2019;10:769.
38. Recordon-Pinson P, Alves BM, Tumiotto C, Bellecave P, Bonnet F, Neau D, et al. A new HIV-1 circulating recombinant form (CRF98_cpx) between CRF06_cpx and subtype B identified in Southwestern France. AIDS Res Hum Retroviruses. 2018;34:1005-9.
39. Patiño-Galindo JÁ, Domínguez F, Cuevas MT, Delgado E, Sánchez M, Pérez-Álvarez L, et al. Genome-scale analysis of evolutionary rate and selection in a fast-expanding Spanish cluster of HIV-1 subtype F1. Infect Genet Evol. 2018;66:43-7.
40. Pérez-Parra S, Chueca N, Álvarez M, Pasquau J, Omar M, Collado A, et al. High prevalence and diversity of HIV-1 non-B genetic forms due to immigration in southern Spain:a phylogeographic approach. PLoS One. 2017;12:0186928.
41. Update on the Transition to Dolutegravir-Based Antiretroviral Therapy:Report of a WHO Meeting. 1st ed. Geneva:World Health Organization;2022.
42. NNRTI Resistance Comments – HIV Drug Resistance Database. Available from:https://hivdb.stanford.edu/dr-summary/comments/NNRTI [Last accessed on 2025 Jan 05].
43. Onywera H, Maman D, Inzaule S, Auma E, Were K, Fredrick H, et al. Surveillance of HIV-1 pol transmitted drug resistance in acutely and recently infected antiretroviral drug-naïve persons in rural western Kenya. PLoS One. 2017;12:0171124.
44. Ceccarelli L, Salpini R, Moudourou S, Cento V, Santoro MM, Fokam J, et al. Characterization of drug resistance mutations in naïve and ART-treated patients infected with HIV-1 in Yaounde, Cameroon. J Med Virol. 2012;84:721-7.
45. Tchiakpe E, Keke RK, Vidal N, Ahoussinou C, Sekpe O, Dagba HG, et al. Moderate rate of transmitted resistance mutations to antiretrovirals and genetic diversity in newly HIV-1 patients diagnosed in Benin. BMC Res Notes. 2020;13:314.
46. YaotsèDA, Nicole V, Roch NF, Mireille PD, Eric D, Martine P. Genetic characterization of HIV-1 strains in Togo reveals a high genetic complexity and genotypic drug-resistance mutations in ARV naive patients. Infect Genet Evol. 2009;9:646-52.
47. Tebit DM, SangaréL, Tiba F, Saydou Y, Makamtse A, Somlare H, et al. Analysis of the diversity of the HIV-1 pol gene and drug resistance associated changes among drug-naïve patients in Burkina Faso. J Med Virol. 2009;81:1691-701.
48. Yebra G, Delgado R, Pulido F, Rubio R, Galán JC, Moreno S, et al. Different trends of transmitted HIV-1 drug resistance in Madrid, Spain, among risk groups in the last decade. Arch Virol. 2014;159:1079-87.
49. Viñuela L, de Salazar A, Fuentes A, Serrano-Conde E, Falces-Romero I, Pinto A, et al. Transmitted drug resistance to antiretroviral drugs in Spain during the period 2019–2021. J Med Virol. 2023;95:29287.
50. Visseaux B, Assoumou L, Mahjoub N, Grude M, Trabaud MA, Raymond S, et al. Surveillance of HIV-1 primary infections in France from 2014 2016:toward stable resistance, but higher diversity, clustering and virulence?J Antimicrob Chemother. 2020;75:183-93.
51. Assoumou L, Bocket L, Pallier C, Grude M, Ait-Namane R, Izopet J, et al. Stable prevalence of transmitted drug resistance mutations and increased circulation of non-B subtypes in antiretroviral-naive chronically HIV-infected patients in 2015/2016 in France. J Antimicrob Chemother. 2019;74: 1417-24.
52. Geremia N, Basso M, De Vito A, Scaggiante R, Giobbia M, Battagin G, et al. Patterns of transmitted drug resistance mutations and HIV-1 subtype dynamics in ART-naïve individuals in Veneto, Italy, from 2017 2024. Viruses. 2024;16:1393.
53. Margot NA, Wong P, Kulkarni R, White K, Porter D, Abram ME, et al. Commonly transmitted HIV-1 drug resistance mutations in reverse-transcriptase and protease in antiretroviral treatment-naive patients and response to regimens containing tenofovir disoproxil fumarate or tenofovir alafenamide. J Infect Dis. 2017;215:920-7.
54. Khodadad N, Hashempour A, Nazar MM, Ghasabi F. Evaluating HIV drug resistance in the middle East and North Africa and its associated factors:a systematic review. Virol J. 2025;22:112.
55. Hamers RL, Rinke de Wit TF, Holmes CB. HIV drug resistance in low-income and middle-income countries. Lancet HIV. 2018;5:588-96.
56. Rizk NA, El Helou R, AlMukdad S, Fahme S, Mumtaz GR, Chemaitelly H, et al. Has UNAIDS left the middle east and north Africa behind?Lancet HIV. 2023;10:630-2.
57. Moyo S, Hunt G, Zuma K, Zungu M, Marinda E, Mabaso M, et al. HIV drug resistance profile in South Africa:findings and implications from the 2017 national HIV household survey. PLoS One. 2020;15:0241071.
58. Ng'ong'a GO, Ayodo G, Kawaka F, Knight V, Ngayo M, Lwembe RM. Antiretroviral (ARV) drug resistance and HIV-1 subtypes among injecting drug users in the coastal region of Kenya. Adv Virol. 2022;2022: 3217749.
59. Assoumou L, Charpentier C, Recordon-Pinson P, GrudéM, Pallier C, Morand-Joubert L, et al. Prevalence of HIV-1 drug resistance in treated patients with viral load >50 copies/mL:a 2014 French nationwide study. J Antimicrob Chemother. 2017;72:1769-73.
60. Miranda MN, Pingarilho M, Pimentel V, Martins MR, Kaiser R, Seguin-Devaux C, et al. Trends of transmitted and acquired drug resistance in Europe From 1981 2019:a comparison between the populations of late presenters and non-late presenters. Front Microbiol. 2022;13:846943.
61. Mimtsoudis I, Tsachouridou O, Akinosoglou K, Metallidis S. Treatment management challenges in naïve and experienced HIV-1-infected individuals carrying the M184V mutation. Viruses. 2024;16:1392.